Currently Empty: $0.00
🧪 Compounds & Molecules
Year 8 Science – Chemistry Unit
📘 KS3 Science
⚗️ Chemistry
🌍 Year 8 UK
🎯 Interactive Lesson
📋 Today's Learning Journey
▶️ Watch First!
Intro Video
▶️
📹 Insert Your Lesson Video Here
Paste your YouTube embed code below<iframe width="100%" height="315" src="YOUR_YOUTUBE_URL"></iframe>
⚛️ Atoms – The Building Blocks
Section 1 – The Smallest Particles
Everything around you — water, air, your skin, this screen — is made of incredibly tiny particles called atoms. An atom is the smallest particle of an element that still has the chemical properties of that element. Atoms are so small that millions of them would fit on the full stop at the end of this sentence.
🔬 Key Definition: An atom is the smallest particle of a substance that can exist. Atoms are made of even smaller sub-atomic particles — a nucleus containing protons and neutrons, surrounded by electrons.
Different elements are made of different types of atoms. For example, a gold ring is made entirely of gold atoms, and oxygen gas contains only oxygen atoms.
⚛️
Protons
Positively charged, in the nucleus
🔵
Neutrons
No charge, in the nucleus
🟢
Electrons
Negatively charged, orbit nucleus
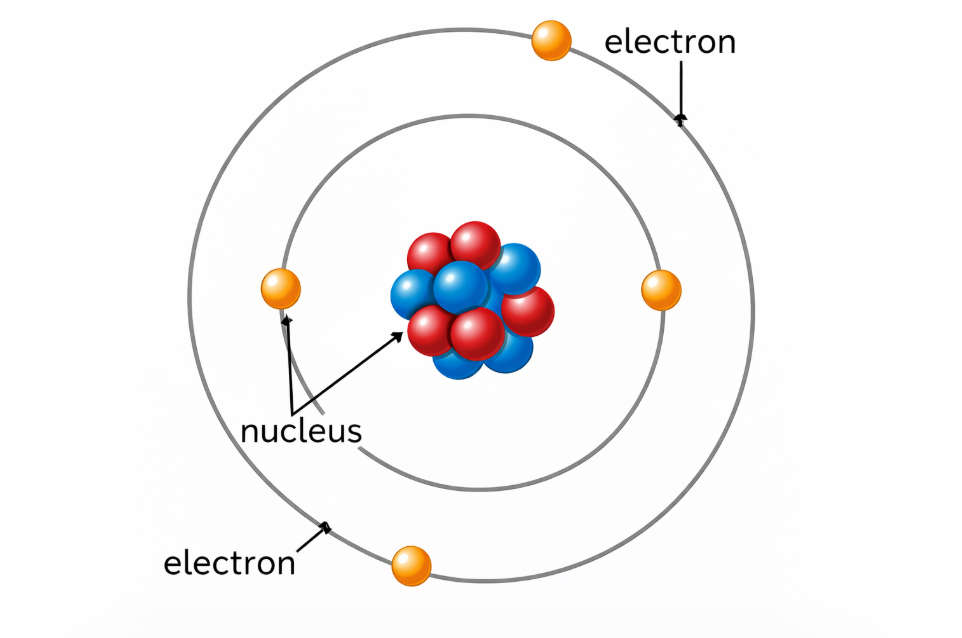
Image: Labelled Diagram of an Atom (protons, neutrons, electrons)
📌 diagram showing the nucleus and electron shells of a simple atom✏️ Quick Check Questions
1 What is an atom?
An atom is the smallest particle of an element that still has the chemical properties of that element. All matter is made of atoms.
2 Name the three sub-atomic particles found in an atom and state their charges.
Protons (positive charge, in nucleus), Neutrons (no charge, in nucleus), Electrons (negative charge, orbit the nucleus).
3 Why is it correct to say that gold jewellery is made of gold atoms?
Gold is a pure element, meaning it contains only one type of atom — gold atoms. Every piece of pure gold contains only gold atoms, which is why it has the properties of gold.
🔵 Elements
Section 2 – Pure Substances Made of One Type of Atom
An element is a pure substance made up of only one type of atom. Elements cannot be broken down into simpler substances by chemical reactions. There are 118 known elements, all listed in the Periodic Table.
📋 The Periodic Table organises all known elements. Each element has a unique chemical symbol — usually 1 or 2 letters. For example: Hydrogen = H, Oxygen = O, Carbon = C, Sodium = Na.
| Element | Symbol | Type | State at Room Temp. |
|---|---|---|---|
| Hydrogen | H | Non-metal | Gas |
| Oxygen | O | Non-metal | Gas |
| Carbon | C | Non-metal | Solid |
| Iron | Fe | Metal | Solid |
| Sodium | Na | Metal | Solid |
| Chlorine | Cl | Non-metal | Gas |
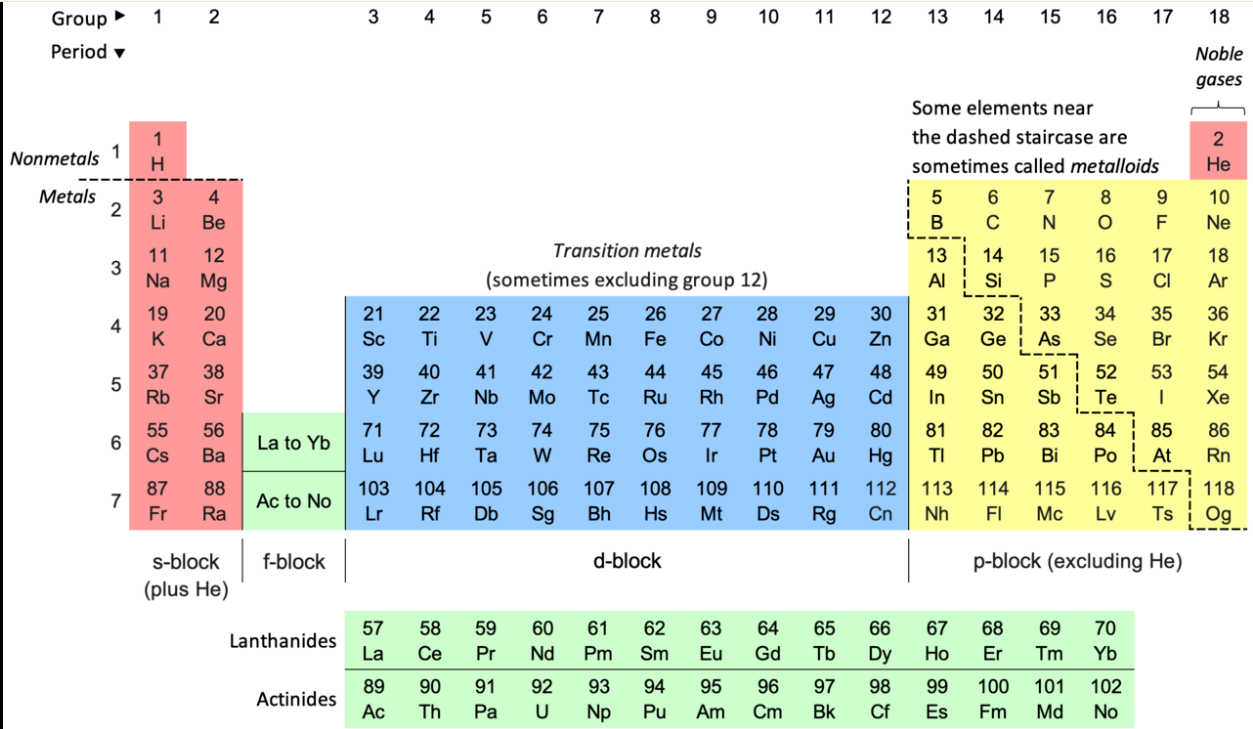
Image: The Periodic Table of Elements
📌 image of the periodic table, highlighting common elements✏️ Quick Check Questions
1 What is an element?
An element is a pure substance made of only one type of atom. It cannot be broken down into simpler substances by chemical reactions. There are 118 known elements.
2 What is the chemical symbol for: (a) Oxygen (b) Carbon (c) Sodium?
(a) Oxygen = O (b) Carbon = C (c) Sodium = Na
3 How many elements are currently known, and where are they all listed?
There are 118 known elements, all listed and organised in the Periodic Table.
💠 Molecules
Section 3 – Two or More Atoms Joined Together
A molecule is formed when two or more atoms are chemically bonded (joined) together. Molecules can be made of atoms of the same element or atoms of different elements.
🔗 Key Idea: When atoms join together, they form a chemical bond. This bond holds the atoms together and creates a molecule with new properties different from the individual atoms.
Examples of molecules made of the same element:
Hhydrogen
+
Hhydrogen
→
H₂ molecule
Examples of molecules made of different elements (these are called compounds — see next section!):
Hhydrogen
+
Hhydrogen
+
Ooxygen
→
H₂O molecule
💧
O₂
Oxygen molecule — 2 oxygen atoms
🟡
N₂
Nitrogen molecule — 2 nitrogen atoms
🌿
Cl₂
Chlorine molecule — 2 chlorine atoms
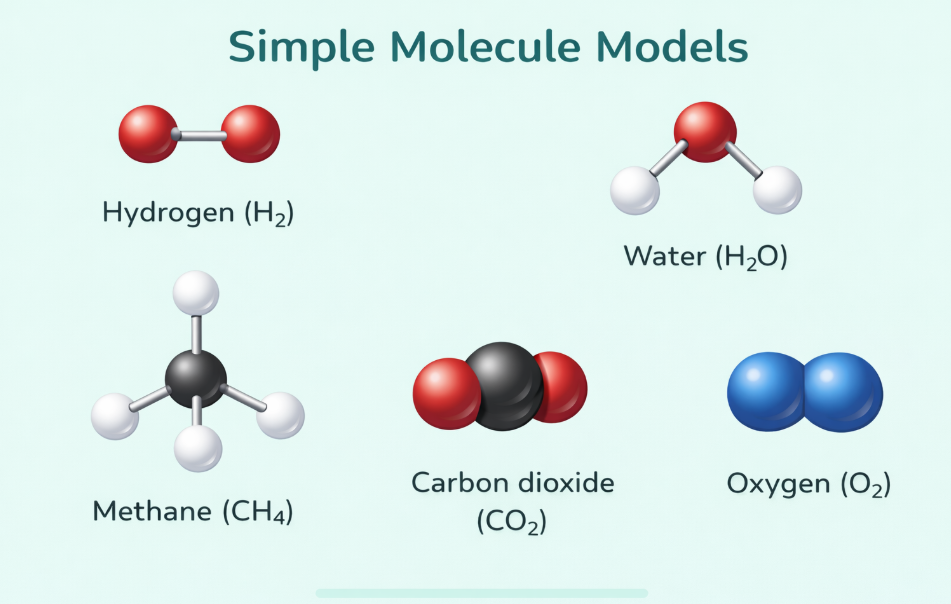
Image: Ball-and-stick models of molecules (H₂, O₂, H₂O)
📌 Add ball-and-stick or space-fill model diagrams of simple molecules✏️ Quick Check Questions
1 What is a molecule?
A molecule is two or more atoms chemically bonded (joined) together. Molecules can be made of atoms of the same element or different elements.
2 An oxygen molecule has the formula O₂. What does this tell you?
O₂ tells us that one molecule of oxygen contains 2 oxygen atoms bonded together. It is made of only one type of atom (oxygen), so it is an element in molecular form.
3 What is the difference between a molecule made of the same atoms and a molecule made of different atoms?
A molecule made of the same atoms (e.g. O₂, H₂) is still a pure element. A molecule made of different types of atoms (e.g. H₂O, CO₂) is called a compound.
🔗 Compounds
Section 4 – Two or More Different Elements Chemically Joined
A compound is a substance made of two or more different elements chemically bonded together. The elements in a compound are joined together in fixed ratios and can only be separated by chemical reactions — not by physical methods like filtering or evaporation.
🌟 Key Definition: A compound is a pure substance containing two or more different elements chemically combined in a fixed ratio. It has completely different properties from the elements it is made from!
A brilliant example: water (H₂O) is a compound of hydrogen and oxygen. Hydrogen is a flammable gas and oxygen helps things burn — yet together they make water, which is used to put fires OUT! The compound is completely different from its elements.
Hydrogen (H) + Oxygen (O) → Water (H₂O)
Sodium (Na) + Chlorine (Cl) → Sodium Chloride (NaCl) — table salt!
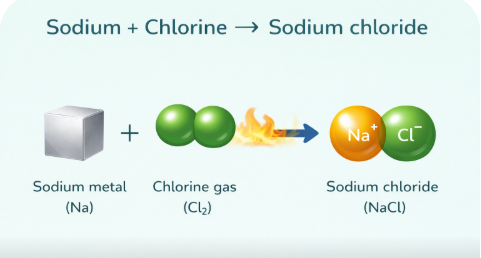
Image: Sodium + Chlorine Reaction forming Salt (NaCl)
📌 diagram showing the reaction of sodium metal + chlorine gas → sodium chloride✏️ Quick Check Questions
1 What is a compound?
A compound is a pure substance made of two or more different elements chemically bonded together in fixed ratios. Compounds can only be separated by chemical reactions.
2 Why is water a surprising compound, given the properties of its elements?
Hydrogen is a flammable gas and oxygen supports burning, yet water (H₂O) puts out fires. This shows that a compound has completely different properties from the elements it is made from.
3 How can you separate the elements in a compound?
The elements in a compound can only be separated by chemical reactions (not physical methods like filtering or evaporation). For example, electrolysis can split water into hydrogen and oxygen.
✏️ Chemical Formulae
Section 5 – Writing and Reading Chemical Formulae
A chemical formula tells us exactly which elements are in a compound and how many atoms of each element are present in one molecule of that compound. It uses element symbols and subscript numbers.
📝 How to read a formula: In H₂O — H is hydrogen, O is oxygen, and the subscript 2 means there are TWO hydrogen atoms. No subscript after a symbol means just ONE atom.
| Compound | Formula | Elements | Atoms per molecule |
|---|---|---|---|
| Water | H₂O | Hydrogen, Oxygen | 2H + 1O = 3 atoms |
| Carbon dioxide | CO₂ | Carbon, Oxygen | 1C + 2O = 3 atoms |
| Sodium chloride | NaCl | Sodium, Chlorine | 1Na + 1Cl = 2 atoms |
| Methane | CH₄ | Carbon, Hydrogen | 1C + 4H = 5 atoms |
| Ammonia | NH₃ | Nitrogen, Hydrogen | 1N + 3H = 4 atoms |
| Glucose | C₆H₁₂O₆ | Carbon, Hydrogen, Oxygen | 6C + 12H + 6O = 24 atoms |
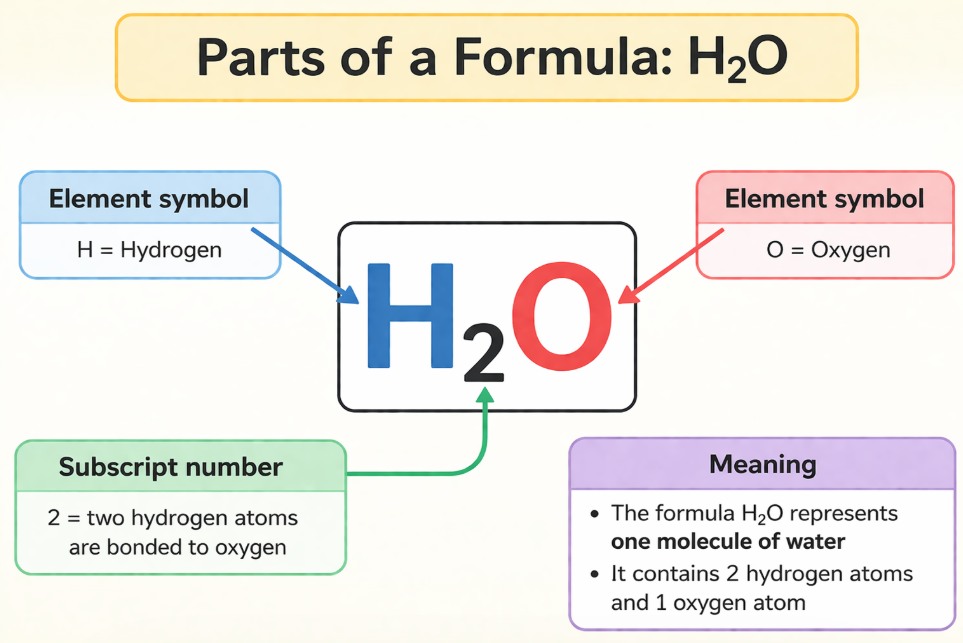
Image: How to Read a Chemical Formula (annotated diagram)
📌 diagram labelling the parts of a formula e.g. H₂O with arrows to symbol, subscript number
✏️ Naming compounds: Many compound names tell you what elements they contain. For example — sodium chloride contains sodium and chlorine. Carbon dioxide contains carbon and oxygen ("di" = two oxygen atoms). Iron oxide contains iron and oxygen.
✏️ Quick Check Questions
1 What information does a chemical formula give us?
A chemical formula tells us which elements are present in a compound and how many atoms of each element are in one molecule of that compound.
2 Explain what CO₂ tells us about carbon dioxide.
CO₂ tells us that one molecule of carbon dioxide contains 1 carbon atom (C) and 2 oxygen atoms (O₂). It is a compound made of two different elements: carbon and oxygen.
3 How many atoms are in one molecule of methane (CH₄)? Name each element present.
One molecule of methane has 5 atoms total: 1 carbon atom (C) and 4 hydrogen atoms (H₄). The elements present are carbon and hydrogen.
🌀 Compounds vs Mixtures
Section 6 – A Very Important Difference!
Students often confuse compounds and mixtures. They are very different things. In a mixture, substances are simply mixed together without a chemical reaction — the substances keep their own properties and can be separated by physical methods.
| Feature | Compound ✅ | Mixture ⚡ |
|---|---|---|
| How formed? | Chemical reaction (elements join) | Physical mixing (no reaction) |
| Properties | Different from its elements | Mix of properties of each substance |
| Fixed ratio? | Yes — always the same ratio | No — any amounts can be mixed |
| Separation? | Only by chemical reaction | Physical methods (filtering, distillation…) |
| Example | Water (H₂O) — always 2H:1O | Air — varying amounts of N₂, O₂, CO₂ |
⚠️ Common Mistake! Air is a mixture of gases (mainly nitrogen, oxygen and carbon dioxide) — NOT a compound. The gases in air are not chemically bonded and can be separated.
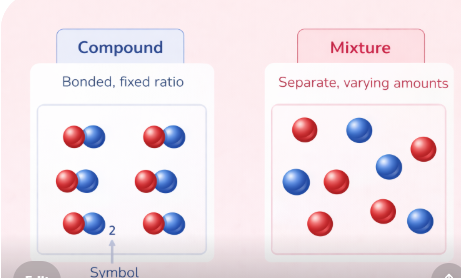
Image: Compound vs Mixture – Particle Diagram Comparison
📌 Add particle diagrams showing atoms in a compound (bonded, fixed ratio) vs a mixture (separate, varying amounts)✏️ Quick Check Questions
1 State TWO differences between a compound and a mixture.
1) A compound is formed by a chemical reaction; a mixture is formed by physical mixing with no chemical reaction. 2) A compound has fixed ratios of elements; a mixture can have any amounts mixed together. 3) A compound can only be separated by chemical reactions; a mixture can be separated by physical methods.
2 Is air a compound or a mixture? Explain your answer.
Air is a mixture. It contains several gases (mainly nitrogen ~78%, oxygen ~21%, carbon dioxide and others) that are not chemically bonded. They keep their own properties and can be separated by physical methods such as fractional distillation.
3 Salt dissolved in water — is this a compound or a mixture? Give a reason.
Salt dissolved in water (salt water) is a mixture. The salt and water have not chemically reacted — they are just mixed together. They can be separated by evaporation (a physical method).
🧬 Common Compounds in Everyday Life
Section 7 – Chemistry All Around Us
Compounds are everywhere in daily life. Understanding their formulae and properties helps us understand the world around us — from the water we drink to the air we breathe.
💧
H₂O – Water
2H + 1O | Essential for life
🌬️
CO₂ – Carbon Dioxide
1C + 2O | Greenhouse gas, breathed out
🧂
NaCl – Sodium Chloride
1Na + 1Cl | Table salt
🧪
HCl – Hydrochloric Acid
1H + 1Cl | Found in stomach acid
🔥
CH₄ – Methane
1C + 4H | Natural gas fuel
🌿
NH₃ – Ammonia
1N + 3H | Used in fertilisers
🍬
C₆H₁₂O₆ – Glucose
6C + 12H + 6O | Sugar, used in respiration
💊
CaCO₃ – Calcium Carbonate
1Ca + 1C + 3O | Limestone, chalk
🧪 Did you know? Rust is a compound too! It forms when iron reacts with oxygen and water. Its formula is Fe₂O₃ — iron oxide. This is why iron objects left outside go rusty!
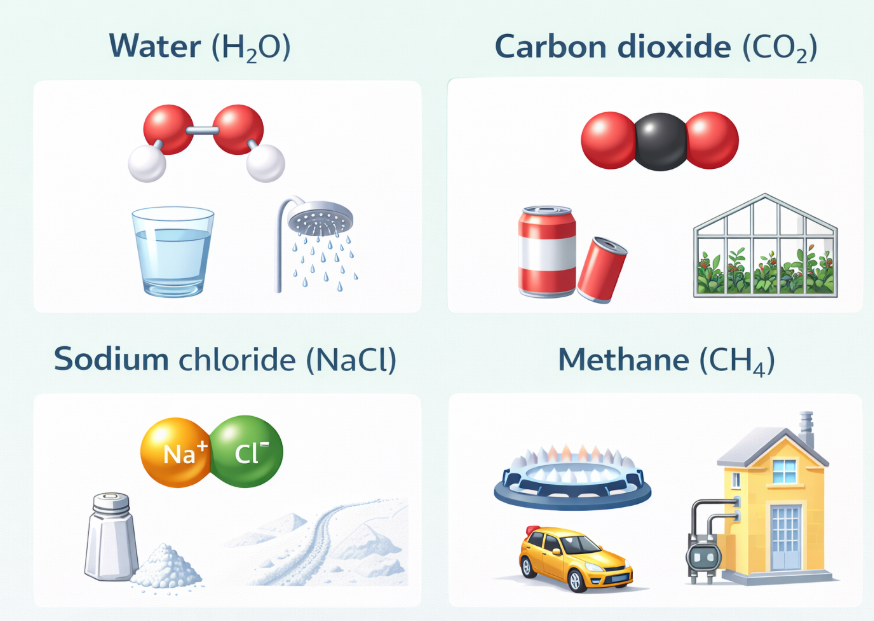
Image: Common Compounds and Their Uses in Daily Life
📌 infographic showing H₂O, CO₂, NaCl, CH₄ etc. with their uses✏️ Quick Check Questions
1 What is the chemical formula for: (a) water (b) carbon dioxide (c) sodium chloride?
(a) Water = H₂O (b) Carbon dioxide = CO₂ (c) Sodium chloride = NaCl
2 What is rust and how does it form? Give its chemical formula.
Rust is iron oxide (Fe₂O₃), a compound formed when iron reacts with oxygen and water. It is a chemical reaction — the iron, oxygen and water chemically combine to form a new substance with different properties.
3 Name the elements present in glucose (C₆H₁₂O₆) and state the number of atoms of each.
Glucose contains three elements: Carbon (6 atoms), Hydrogen (12 atoms), and Oxygen (6 atoms). One molecule of glucose has 24 atoms in total.
📝 Multiple Choice Quiz
Section 8 – 10 Questions
🎯 MCQ Challenge
Select the best answer for each question, then submit!
0 of 10 answered
✍️ Long Answer Questions
Section 9 – Extended Writing Practice
📖 Extended Writing
Write your detailed answers — then reveal the model answer & mark scheme!
🔤 One Word Quiz
Section 10 – 20 Questions
⚡ One Word Challenge
Type your answer and click Check — or press Enter!