Currently Empty: $0.00
Lesson 14 : Periodic Table
Year 8 • Chemistry
Lesson: The Periodic Table – An Introduction
In this lesson, students learn what the periodic table is, how elements are arranged,
and why elements in the same group have similar properties. The lesson introduces metals,
non-metals, and key element groups studied in Year 8.
Lesson Objectives
- Understand what the periodic table shows.
- Explain how elements are arranged.
- Identify groups and periods.
- Describe metals, non-metals, and metalloids.
- Recall properties of Group 1, Group 7, and Group 0.
1. What Is the Periodic Table?
The periodic table is a chart that shows all known elements arranged in a specific order.
Each element is shown using a symbol and an atomic number.
-
Helps us predict properties of elements
Each element is shown using:
-
Symbol (e.g. H, O, Na)
-
Atomic number (number of protons)
-
Name of the element
📜 A Short History
-
In 1869, a scientist named Dmitri Mendeleev arranged elements by atomic mass
-
He noticed repeating patterns in properties
-
He left gaps for undiscovered elements
-
Modern periodic table is arranged by atomic number
👉 This repeating pattern is called periodicity
-
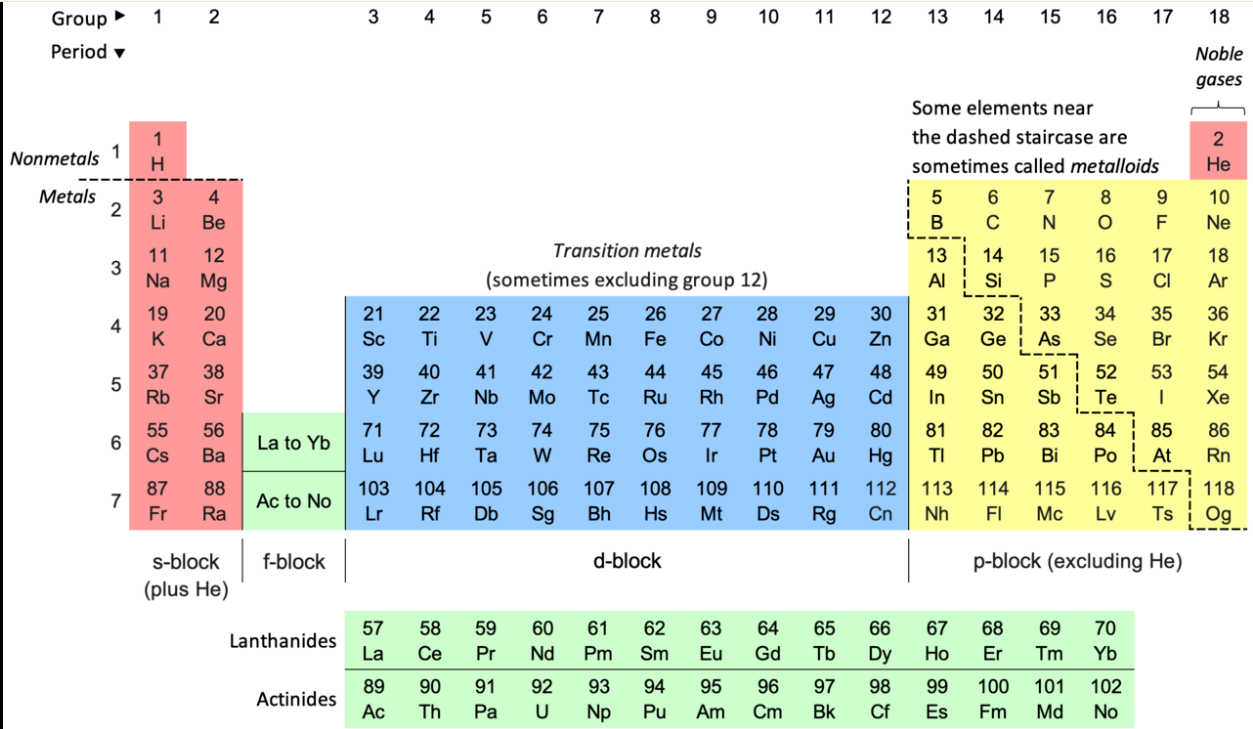
Key ideas:
• Each box represents one element
• Atomic number = number of protons
• Elements are arranged in a logical order
Real-life example: Oxygen, carbon, and iron are all elements found in the periodic table.
2. Groups and Periods
The periodic table is organised into groups and periods.

• Groups are vertical columns
• Elements in the same group have similar properties
-
Period number = number of electron shells
-
Example:
-
Period 2 elements have 2 electron shells
-
• Periods are horizontal rows
• Period number shows number of electron shells
-
Have similar properties
-
Have the same number of outer shell electrons
Example: All Group 1 elements are reactive metals.
3. Metals, Non-Metals, and Metalloids
Elements can be classified based on their properties.

Metals:
-
Found on the left and middle
-
Properties:
-
Shiny
-
Good conductors of heat & electricity
-
Malleable (can be hammered)
-
Examples: Iron, Copper, Aluminium
Non-metals:
-
Found on the right
-
Properties:
-
Dull
-
Poor conductors
-
Often gases
-
Examples: Oxygen, Carbon, Nitrogen
Metalloids: properties of both metals and non-metals
-
Found along the zig-zag line
-
Have properties of both metals and non-metals
Example: Silicon
Example: Iron is a metal, oxygen is a non-metal, silicon is a metalloid.
4. Important Groups in Year 8
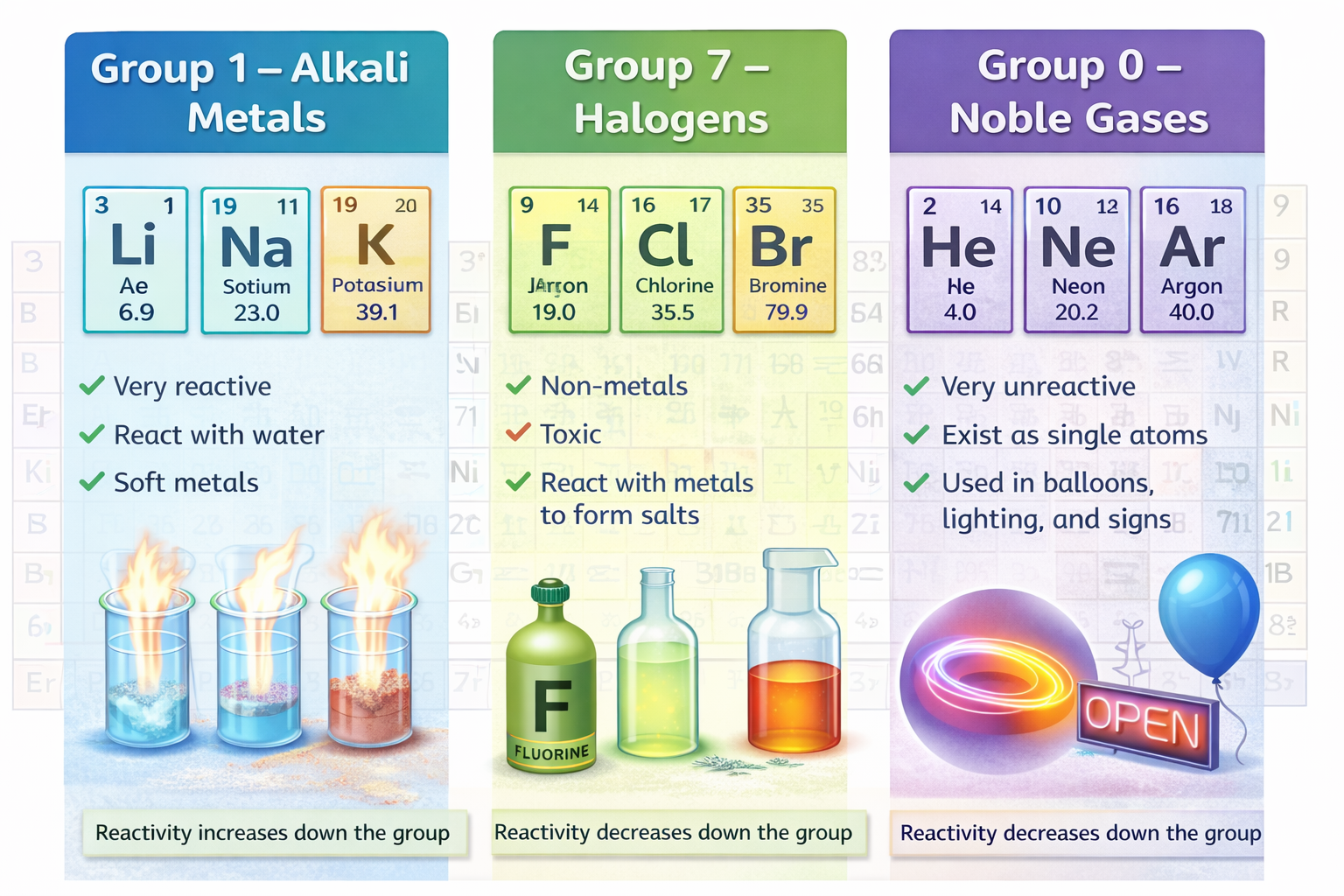
Group 1 – Alkali Metals: very reactive, react with water
Examples: Lithium, Sodium, Potassium
Properties:
-
Very reactive
-
React with water
-
Soft metals
-
Stored in oil
Reactivity increases down the group
Group 7 – Halogens: reactive non-metals, form salts
Examples: Fluorine, Chlorine, Bromine
Properties:
-
Non-metals
-
Toxic
-
React with metals to form salts
Reactivity decreases down the group
Group 0 – Noble Gases: very unreactive gases
Examples: Helium, Neon, Argon
Properties:
-
Very unreactive
-
Exist as single atoms
-
Used in balloons, lighting, and signs
✅ Quick Summary
-
The periodic table organises elements
-
Rows = periods
-
Columns = groups
-
Elements in the same group behave similarly
-
Metals, non-metals, and metalloids have different properties
-
Practice Questions
A. Fill in the Blanks
- The periodic table lists all known __________.
- Vertical columns are called __________.
- Horizontal rows are called __________.
- Group 0 elements are very __________.
- Metals are good __________ of electricity.
B. True or False
- Elements in the same group have similar properties.
- All elements are metals.
- The atomic number shows number of protons.
- Group 1 metals are unreactive.
- Noble gases are found in Group 0.
C. Short Answer
- What is the periodic table?
- What is a group?
- Name one property of metals.
- Why are noble gases unreactive?
- Name one Group 7 element.
✅ Show Answer Key
A. Fill in the Blanks
- elements
- groups
- periods
- unreactive
- conductors
B. True or False
- True
- False
- True
- False
- True
C. Short Answer
- A table that organises all elements.
- A vertical column of elements.
- Shiny / conducts electricity.
- They have full outer shells.
- Chlorine / Bromine / Fluorine.
© Aviate Learning – The Periodic Table (Year 8 Chemistry)

