Currently Empty: $0.00
Lesson 29: Combustion Reactions What is combustion? Fuels and oxygen Fire triangle
Year 8 • Chemistry
🔥 Combustion Reactions
Learn what combustion reactions are, how fuels burn in oxygen, the fire triangle,
and the difference between complete and incomplete combustion — with examples and practice.
Lesson Objectives
- Understand what a combustion reaction is.
- Identify fuels and products of combustion.
- Explain the fire triangle.
- Distinguish between complete and incomplete combustion.
- Write simple word and symbol equations.

Examples of combustion: candle, campfire, gas stove and fuels
🔳 Image Placeholder — Burning candle / campfire / fuel examples
1. What is a Combustion Reaction?
A combustion reaction is a chemical reaction in which a substance reacts with oxygen and releases energy (usually as heat and light).
Combustion = Burning
- A fuel (something that burns)
- Oxygen (from the air)
2. Key Features of Combustion
- Need oxygen
- Produce energy (heat and/or light)
- Form new substances (products)
3. Common Fuels That Burn
| Fuel Type | Examples |
|---|---|
| Solids | Wood, coal, charcoal |
| Liquids | Petrol, diesel, ethanol |
| Gases | Methane, propane, butane |
4. Products of Combustion
Hydrocarbons → Carbon dioxide + Water
Methane + Oxygen → Carbon dioxide + Water
CH₄ + 2O₂ → CO₂ + 2H₂O
Metals → Metal oxides
Magnesium + Oxygen → Magnesium oxide
2Mg + O₂ → 2MgO
Iron + Oxygen → Iron oxide
5. Exothermic Reactions
- Combustion reactions are exothermic.
- They release energy to the surroundings.
- A candle gives out heat and light.
Examples of combustion: candle, campfire, gas stove and fuels
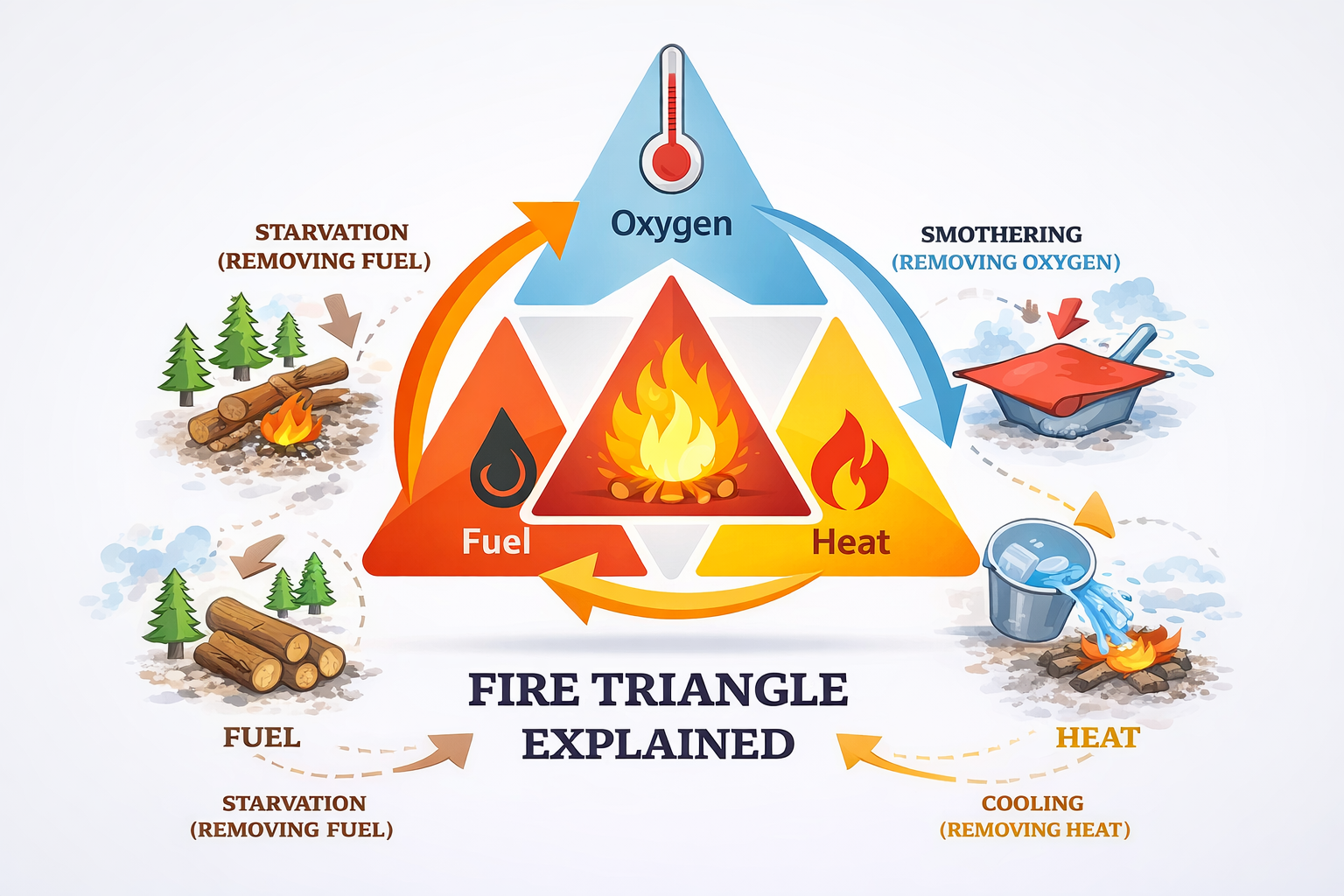
6. The Fire Triangle
- Fuel
- Oxygen
- Heat
If any one is removed, the fire goes out.
7. Complete and Incomplete Combustion
Complete combustion:
Enough oxygen → CO₂ + H₂O
Enough oxygen → CO₂ + H₂O
CH₄ + 2O₂ → CO₂ + 2H₂O
Incomplete combustion:
Not enough oxygen → CO, soot, H₂O
Not enough oxygen → CO, soot, H₂O
2CH₄ + 3O₂ → 2CO + 4H₂O
⚠️ Carbon monoxide is toxic.
8. Real-Life Examples
- Candle → Wax → CO₂ + H₂O
- Car → Petrol → CO₂ + H₂O
- Gas stove → Methane → CO₂ + H₂O
- Campfire → Wood → CO₂ + H₂O + ash
9. Writing Word & Symbol Equations
Hydrogen + Oxygen → Water
2H₂ + O₂ → 2H₂O
✏️ Worked Examples
- Magnesium + Oxygen → Magnesium oxide
- Propane + Oxygen → Carbon dioxide + Water
🧠 Practice Questions
- What is combustion?
- Name two things needed for combustion.
- Is combustion exothermic or endothermic?
- Write word equations for burning hydrogen, ethanol, magnesium.
- Is a yellow smoky flame complete or incomplete combustion?
- Name the three parts of the fire triangle.
- What happens if oxygen is removed?
- Write word equations for burning iron and butane.
✅ Show Answer Key
Answers:
- Combustion = burning in oxygen.
- Fuel and oxygen.
- Exothermic.
- Hydrogen + Oxygen → Water
- Incomplete combustion.
- Fuel, oxygen, heat.
- The fire goes out.
- Iron + Oxygen → Iron oxide
© Aviate Learning – Combustion Reactions (Year 8)

