Currently Empty: $0.00
Year 8 Chemistry
Oxidation and Displacement Reactions
Understanding Oxidation, Displacement and Reactivity Series
🎥 Lesson Introduction
Lesson Objectives
- Understand what oxidation means
- Identify oxidation reactions
- Understand displacement reactions
- Use the reactivity series to predict reactions
- Write word equations and symbol equations
1. Oxidation
What is Oxidation?
In Year 8 chemistry, oxidation means gaining oxygen.
When a substance reacts with oxygen and forms an oxide, oxidation has taken place.
This usually happens when metals react with oxygen in air.
General Pattern
Element + Oxygen → Metal Oxide

Burning Magnesium – Oxidation Reaction
Example 1
Word Equation
Magnesium + Oxygen → Magnesium Oxide
Symbol Equation
2Mg + O₂ → 2MgO
Explanation
Magnesium atoms combine with oxygen molecules to form magnesium oxide. Magnesium gains oxygen, so magnesium is oxidised.
Example 2 (Rusting)
Word Equation
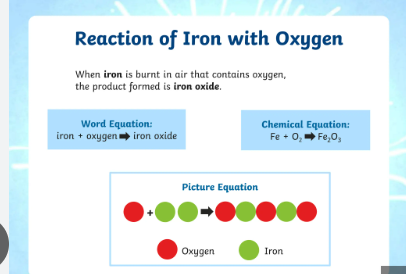
Iron + Oxygen → Iron Oxide
Symbol Equation
4Fe + 3O₂ → 2Fe₂O₃
Explanation
When iron reacts slowly with oxygen and water in air, rust forms. Rust is iron oxide, so iron has been oxidised. Rusting is a slow oxidation reaction.
Rusting of iron
Example 3
Word Equation
Copper + Oxygen → Copper Oxide
Symbol Equation
2Cu + O₂ → 2CuO
Explanation
Copper reacts with oxygen when heated and forms black copper oxide.

Reaction of Copper with Oxygen
Memory Tip
Oxidation = Oxygen Added
Practice Questions
Question 1
Define oxidation.
Show Answer
Answer: Oxidation is a chemical reaction in which a substance gains oxygen.
Explanation: When oxygen combines with another substance to form an oxide, oxidation occurs.
Question 2
Complete the word equation:
Zinc + Oxygen → ______
Show Answer
Answer: Zinc Oxide
Explanation: Zinc reacts with oxygen to form zinc oxide.
Question 3
Write the symbol equation:
Magnesium + Oxygen → Magnesium Oxide
Show Answer
Answer: 2Mg + O₂ → 2MgO
Explanation: The equation is balanced with equal atoms on both sides.
Question 4
Which substance is oxidised?
2Cu + O₂ → 2CuO
Show Answer
Answer: Copper
Explanation: Copper gains oxygen, therefore it is oxidised.
3. Displacement Reactions
A displacement reaction occurs when a more reactive metal replaces a less reactive metal from its compound.

Zinc Displacing Copper from Copper Sulfate
Example
Word Equation
Zinc + Copper Sulfate → Zinc Sulfate + Copper
Symbol Equation
Zn + CuSO₄ → ZnSO₄ + Cu
Explanation
Zinc is more reactive than copper, so zinc replaces copper in copper sulfate. Copper metal is produced.
4. Reactivity Series
Metals can be arranged in order of reactivity.

Reactivity series
Sodium
Calcium
Magnesium
Aluminium
Zinc
Iron
Copper
Silver
Gold
Important Rule
A metal higher in the reactivity series can displace metals below it.
Final Summary
Oxidation — Gain of oxygen
Example:
2Mg + O₂ → 2MgO
Displacement Reaction
A more reactive metal replaces a less reactive metal.
Example:
Zn + CuSO₄ → ZnSO₄ + Cu
Reactivity Rule
Higher metals in the reactivity series displace lower metals.